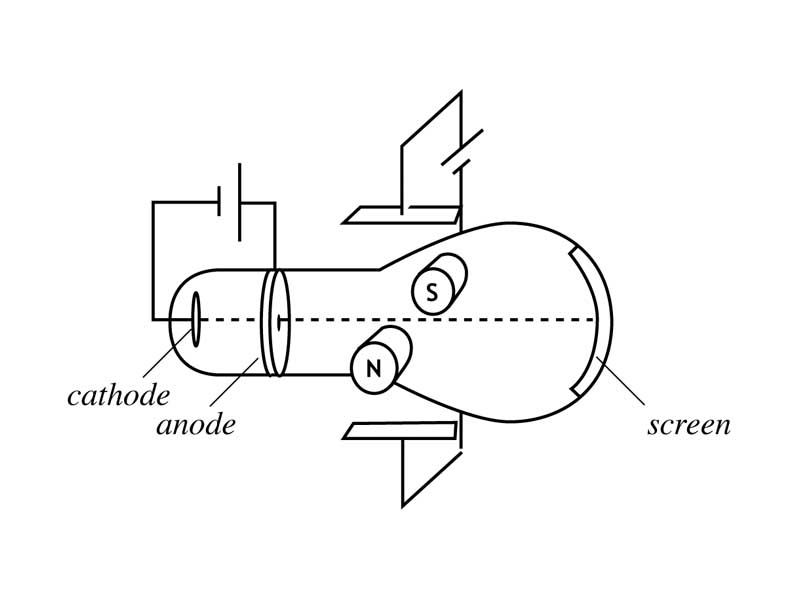 
It was named Cathode Rays because it originated from the Cathode of the Tube.Īfter that, using a perforated Cathode (Cathode with fine pores), he conducted another experiment. The light beam was found to curve towards the positive plate and was thus charged negatively. Then, the light emitted from the ray was passed via the strong electric field formed between two plates, charge positive and negative. Also, he noticed a certain beam of light starting to emanate from the tube's cathode, and this travelled throughout the tube upon reducing the pressure further. It is the kernel or nucleus of the gas that is used in the tube, and thus, it has different properties to that of the cathode rays, made up of electrons.ĭifferences between Cathode and Anode Raysīasically, in the first Canal ray experiment, William used the Crookes tube supplying high voltage and gradually reduced the pressure within the tube chamber from 0.01 to 0.001 atm. When a higher voltage is applied, the experiment ionizes the gas, and it is the positive ions of gas that constitute the canal ray. These rays moved in the opposite direction facing the cathode rays and were called canal rays. When the voltage was increased to thousands of volts, a faint luminous ray was seen, extending from the holes present behind the cathode. Thereby, the electricity starts to flow as the circuit is complete. The high voltage is passed to the two metal pieces to ionize the air by making it an electricity conductor. Let us discuss more details about the procedure of the experiment, as listed below.Īs the apparatus is set up by evacuating the air and giving a high voltage source for maintaining a low pressure inside the tube. The air evacuation lowers the pressure of the gas present inside the tube. 
These two metal pieces are further connected with an external voltage. The apparatus of this experiment includes the same cathode-ray experiment, made up of a glass tube containing two metal ion pieces at different ends that acts as an electrode. Also, a faint luminous ray was seen extending from the holes of the back of the cathode. In this experiment, Goldstein happened to apply a high voltage across a discharge tube that had a perforated cathode. The proton discovery has happened after the electron discovery has further strengthened the structure of the atom. The canal rays experiment is the one that led to the discovery of the proton. He was also one of the first spectrometers to use such ions’ sources. So, it is said that Dempster is the one who discovered canal rays. Thomson led to the mass spectrometry development. Later on, anode rays work by the scientist Wilhelm Wien and J. These rays were observed in 1886 in Crookes tubes when the German scientist named "Eugen Goldstein performed experiments." A Canal ray (also known as a positive or anode ray) is described as a positive ions' beam, created by certain gas-discharge tube types.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
